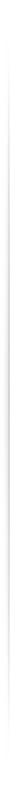|
|
Intravenous Device Manufacturing
Warden Systems is developing a line of Intravenous devices for drug delivery and monitoring equipment to create a feedback mechanism for control. These devices will contain new materials for toxin filtration and sterilization as well as precise measurement sensors to determine how much dose is administered to the patient with a lower chance of error. We also plan to provide these devices on a subscription service to allow smaller, less-funded hospitals obtain the latest devices at a considerably smaller fee compared to other manufacturers.
|
|
Subdural Electrode Brain-Machine Interface
The Biomedical Research lab at Warden Systems works closely with NIH NINDS to process signals coming from subdural electrodes placed in the brains of patients suffering from epilepsy. Modalities such as fMRI are too costly and cannot be adapted into a wearable device, and EEG/MEG have artifacts because of the scaffolding on the cortices. This is a strictly research oriented effort to help the patients and, in the process, understand how the brain works. |
|
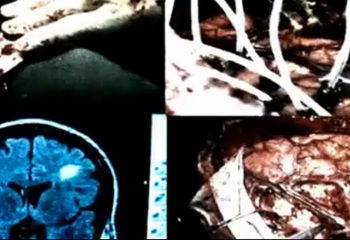
Implantable Medical Devices
We are developing new ferromagnetic implants that have the potential of inducing angiogenesis, as most of the blood contains ferritin complexes which would be attracted to the implant. The implants will be made using a porous composite of hydroxyapatite and magnetized steel on top of a frame of light-weight cobalt-chromium-molybdenum produced by our patented machining technique. The porous composite allows for blood vessels to permeate the structure keeping the surrounding bone healthy and promote osteogenesis. This will hopefully have fewer inflammatory issues, less to no rejection from the body and a longer life-cycle of the implant insuring that the customers of our orthopedic surgery clients have the most return on investment. As of yet, the exploratory material is being tested for Biocompatibility, leading into animal studies, compliant with the Class 3 device requirement for FDA approval. |